As published in the British Journal of Nursing VOL. 28, NO. 15
Neuromuscular electrical stimulation is a developing technology in healthcare that works to improve the condition and strength of muscles. More recently, it has been used within pelvic healthcare to improve and maintain the strength of the pelvic floor muscles to avoid pelvic floor disorders. The below article was written by Brand Coordinator, Eleanor Allon, to introduce the concept of neuromuscular electrical stimulation in relation to the pelvic floor to healthcare professionals and discuss whether it is a practice to move forward with.
Neuromuscular electrical stimulation (NMES) involves using an electrical impulse or signal to elicit a muscle contraction (Knutson et al, 2015). Also known as electrical muscle stimulation (EMS) or electromyostimulation, NMES can be used to:
- Provide strength training for muscles
- Rehabilitate muscles following injury or surgery
- Exercise muscles in a controlled manner to avoid over-exercising
- Mobilise weak or wasted muscles (muscle atrophy)
- Test muscle strength and capacity.
The application of electricity in rehabilitation is not new (Heidland et al, 2013). Live electric fish were studied as an analgesic in around 201 BC by the Greek physician Galen. Some 2000 years later, Genevan physics professor Jean Jallabert used electrical currents in a clinical environment to enhance the quality of the extensor muscles in one patient’s forearm, improving movement significantly as a result (Bussel, 2015).
In the 21st century, electrical stimulation is used to provide strength training to athletes and aid in muscle rehabilitation. This focus on rehabilitation has gained relevance for those seeking to treat pelvic floor dysfunctions, both in a medical setting and at home.
The importance of the pelvic floor
The pelvic floor is vital for routine bodily functions. The muscles of the pelvic floor help to control bladder and bowel movements, support pelvic organs, and help to control intimate sensations in both men and women (Pelvic Obstetric and Gynaecological Physiotherapy (POGP), 2015). One in three women will suffer from a pelvic floor dysfunction during their lifetime (Lawrence et al, 2008).
Pelvic floor dysfunction is the inability to correctly tighten and relax the muscles of the pelvic floor (Cleveland Clinic, 2017), which can lead to one or more of the following conditions:
- Urinary incontinence
- Faecal incontinence
- Pelvic organ prolapse
- Pelvic pain
- Discomfort during sexual intercourse in women
- Erectile dysfunction in men.
NMES of the pelvic floor muscles can stimulate muscle activity, retraining the individual muscles and increasing their strength as a result, effectively treating the conditions named above, among others (Veldman et al, 2016). Optimal pelvic floor muscle strength can aid in:
- Reducing urinary incontinence
- Managing mild and moderate pelvic organ prolapse
- Relieving pelvic pain that is a result of pelvic floor muscle tightness
- Increasing intimate sensitivity by encouraging blood flow.
How does NMES exercise the pelvic floor muscles?
A physiotherapist or urology nurse can advise women on the correct exercise technique and document any changes in pelvic floor muscle strength and function throughout treatment.
NMES has been found to work the pelvic floor muscles (Correia et al, 2014). The electrical signals that are produced from an NMES device target nerve fibres and elicit a contraction of the pelvic floor muscles. As a result, it is often used as a strength-training technique for healthy adults and as a rehabilitation tool to increase or preserve muscle function and mass for those individuals with muscle weakness, or patients who are unable to perform voluntary contractions (Veldman et al, 2016).
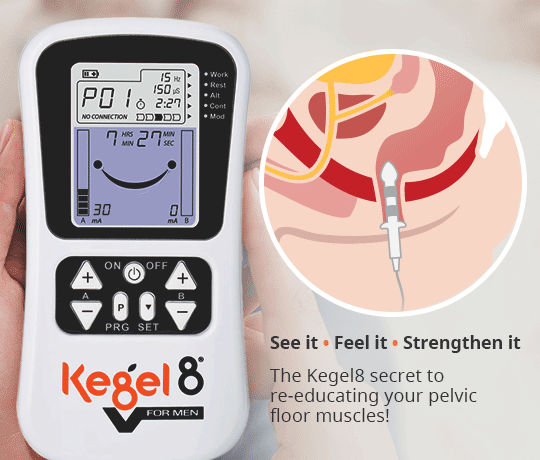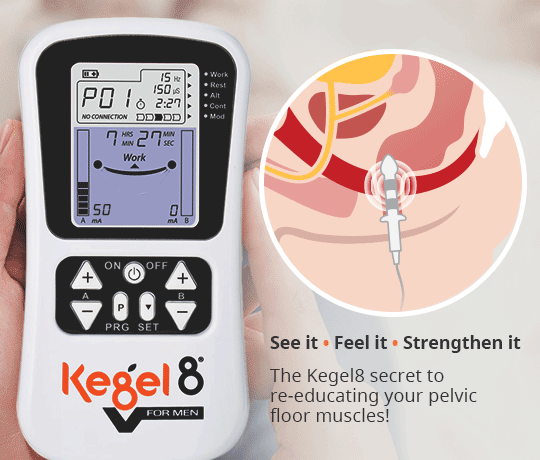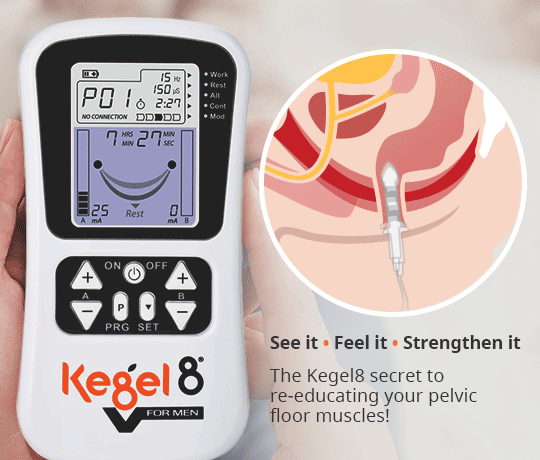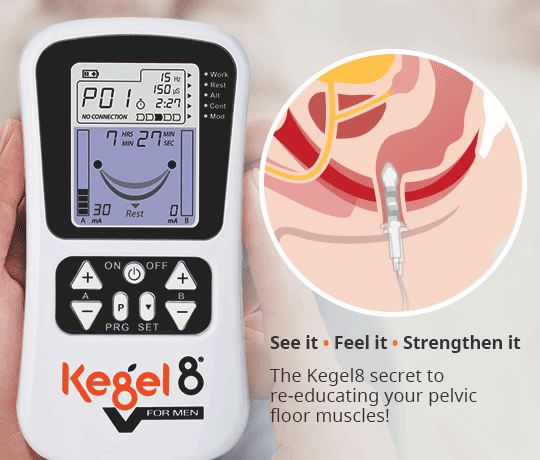
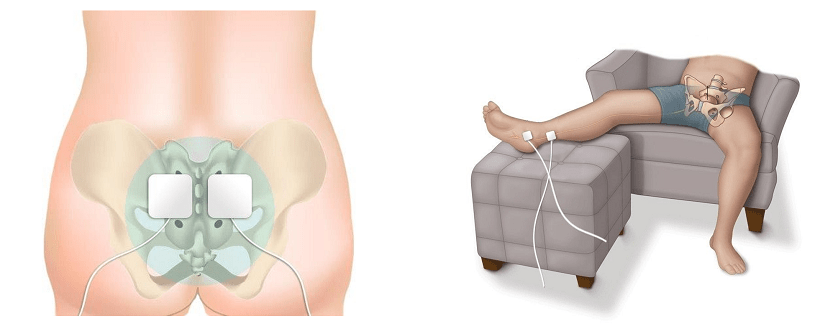
NMES can be applied to the pelvic floor muscles in one of two ways. The first is through a conservative, external application, commonly referred to as surface electrical stimulation (SES) (Correia et al, 2014). This method involves an NMES unit sending impulses to electrode pads that are placed on the skin over nerves on the patient’s body. These electrode pads are often placed to stimulate either the sacral or tibial nerves. To access the sacral nerves, electrode pads can be placed on the patient’s lower back (Figure 1). For the tibial nerve, electrode pads can be placed on the patient’s inner ankle (Figure 2).
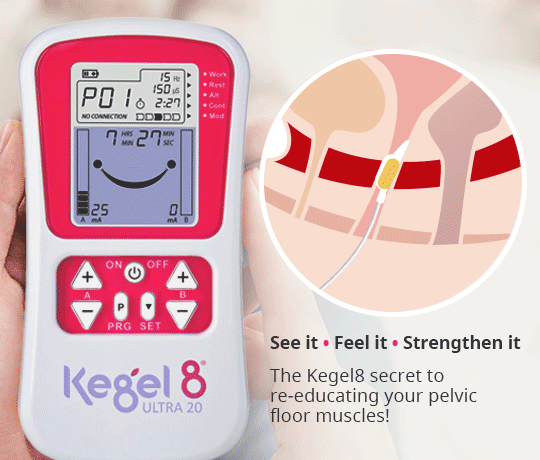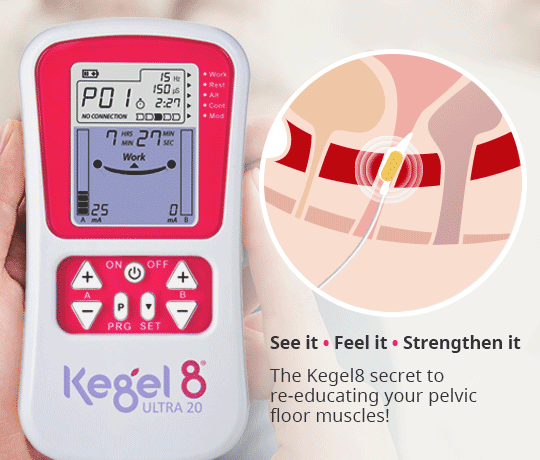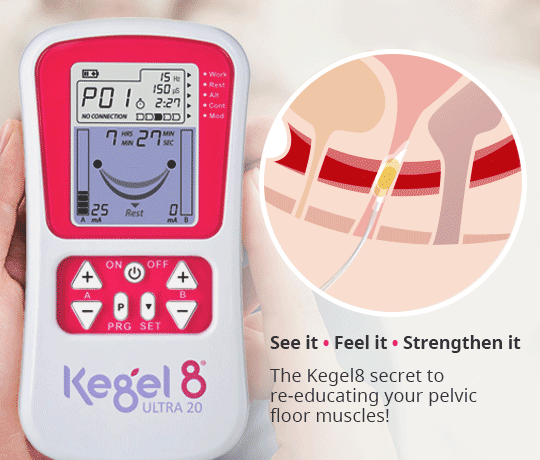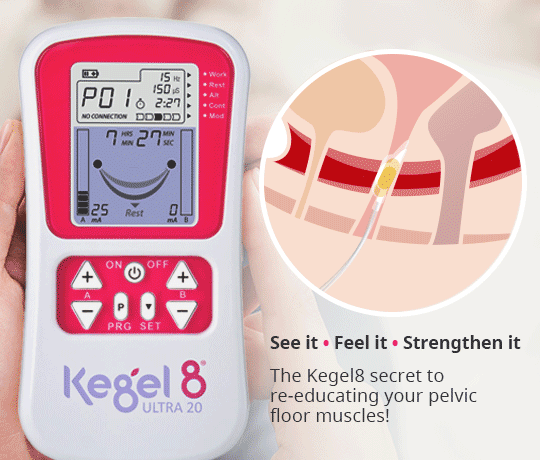
Another method of applying NMES to the pelvic floor muscles is via intravaginal electrical stimulation (IVES). This form of treatment is more widely recognised and is used in clinical practice as a result of its effectiveness, despite the disadvantages of possible patient discomfort and the requirement of electrode sterilisation (Correia et al, 2014). One study reported that nearly 80% of patients recommend probe-based stimulation (Indrekvam and Hunskaar, 2002).
IVES often uses a probe with electrodes on its surface to directly stimulate the pelvic floor muscles. It is these surface electrodes that target the pelvic floor muscles and deliver electrical impulses direct to the treatment area.
Both forms of internal and external electrical stimulation of the pelvic floor muscles recruit two types of nerve fibres to increase muscle function and strength. Lower electrical waveforms can stimulate the slow-twitch nerve fibres—those that are responsible for endurance (and involved in urge incontinence). Higher electrical waveforms tend to stimulate the fast-twitch nerve fibres —those responsible for acting and controlling muscles under a sudden burst of pressure (and involved in stress incontinence) (Herbert, 2003).
According to one study, both SES and IVES are effective in improving pelvic floor dysfunction symptoms, such as urine loss (Correia et al, 2014). However, only intravaginal electrical stimulation was found to increase the strength of pelvic floor muscles and improve muscle function (Correia et al, 2014).
NMES as a treatment for urinary incontinence
NMES devices are used in clinical practice and in the consumer market for treating urinary incontinence. Urinary incontinence is defined as ‘the complaint of any involuntary leakage of urine’ (Abrams et al, 2002). The condition is prevalent in both sexes, but is much more frequent in women (Aoki, 2017). Incontinence in men is often a result of prostatic enlargement or radiotherapy from prostate cancer. In contrast, female incontinence is habitually related to pelvic floor muscle dysfunction, with occurrences of dysfunctions increasing during pregnancy, after childbirth, or during the menopausal process (Aoki et al, 2017). As a result, it is widely considered as unavoidable. Many women do not seek treatment because they are reluctant to talk to their GP about urinary leakage (Diokno et al, 2006).
The two main subtypes of urinary incontinence are stress incontinence and urgency incontinence. Stress incontinence is defined as ‘the complaint of involuntary leakage on effort or exertion, or on sneezing or coughing’ (Abrams et al, 2002). This is a condition that is predominantly caused by the weakening of the muscles that control the discharge of urine from the bladder, causing some urine to escape when the muscles are put under pressure (Royal College of Nursing (RCN), 2016). The POGP group has recognised stress incontinence as the most common form of urinary leakage (POGP, 2017).
Urge incontinence is defined as ‘the complaint of involuntary leakage accompanied by or immediately preceded by urgency’ (Abrams et al, 2002), and can be affected by both diet and the loss of muscle control. For example, caffeine has been shown to have a diuretic effect and increase overactive bladder symptoms (Faiena et al, 2015).
The National Institute for Health and Care Excellence (NICE) recommends a 3-month course of manual pelvic floor muscle training exercises as the first-line treatment for women who experience incontinence and are able to produce an effective pelvic floor muscle contraction without an aid (NICE, 2015). However, more than 30% of women are unable to produce an effective pelvic floor contraction (Bø, 2003), meaning that extra help is required to rehabilitate pelvic floor muscle strength and function.
Electrical stimulation units provide a less invasive, lower risk alternative to surgery. Surgical options for the treatment of urinary incontinence include (NHS website, 2016):
- Colposuspension
- Sling procedures
- Artificial urinary sphincter
- Vaginal mesh tape—use is currently paused (NHS Improvement and NHS England, 2019)
- Catheterisation.
It has been reported that transvaginal pelvic floor electrical stimulation is a safe and effective therapy for genuine stress incontinence, with patient satisfaction rates of more than 50% (Sand et al, 1995).
Efficacy of NMES
Interventions to manage urinary incontinence include: pelvic floor muscle training (PFMT); biofeedback devices; electrical stimulation; and surgery.
As noted earlier, NICE recommends PFMT as the first-line treatment. However, this is specific to women who can already produce an effective pelvic floor muscle contraction. Therefore, for women who struggle to produce a successful contraction, extra intervention is required.
Biofeedback devices, such as vaginal cones, are often used in clinical practice to measure a patient’s pelvic floor muscle strength before proceeding with treatment. These tools have been found to be better than no active treatment, and may be a good, conservative option when used with PFMT (Herbison and Dean, 2013). However, further intervention may be necessary if the patient is still unable to activate their pelvic floor muscles.
The use of NMES as a mode to manage urinary incontinence has been trialled and reviewed for a number of years (Castro et al, 2008; Heidland et al, 2013 Correia et al, 2014; Fürst et al, 2014). Both SES and IVES are important treatments to improve urinary incontinence, particularly stress incontinence (Correia et al, 2014). It has been reported that electrical stimulation can enhance the effect of PFMT, especially in the short term (Berghmans et al, 2013). Results from a 2016 study also suggest that intravaginal NMES is effective in improving pelvic floor muscle tone and treating symptoms of an overactive bladder (Lúcio et al, 2016). More than 55% of patients were satisfied with electrical stimulation as a treatment for stress urinary incontinence (Castro et al, 2008).
Reduction in incontinence pad usage is not only key to improve the quality of life of the patient, but also reduces costs for healthcare providers. In the UK, urinary incontinence and urinary problems cost the NHS more than £26 million per year (Department of Health and Social Care (DHSC), 2016). Incontinence product costs alone total around £80 million per year (NHS England, 2018).
Intravaginal pelvic floor electrostimulation devices are known for their low side-effect profile (Faiena et al, 2015) and are considerably less-invasive than surgical treatments. In a 2013 study, more than 200 women were treated with pelvic floor muscle stimulation to improve stress urinary incontinence. The overall patient satisfaction rate for this modality was found to be 83%, with patients reporting satisfaction with discretion, ease, freedom, and rapidity of use (Chêne et al, 2013).
As for long-term efficacy, a 2014 study reported that nearly 90% of women were satisfied with vaginal electrical stimulation to treat stress urinary incontinence. Nearly 65% were still satisfied 12 months after treatment, and more than 1 in 3 women remained satisfied 96 months post-treatment (Fürst et al, 2014).
Guidelines for using NMES
NMES is still a relatively new treatment method. There is a lack of long-term studies for the efficacy of using NMES to improve urinary and bowel incontinence, therefore guidelines reflect caution when employing the treatment.
NICE has acknowledged that ‘electrical stimulation and/ or biofeedback should be considered for women who cannot actively contract pelvic floor muscles to aid motivation and adherence to therapy’ (NICE, 2019).
There is a great deal of anecdotal evidence to support the use of NMES. As a result, the range of devices that are being marketed as pelvic floor rehabilitation devices by means of enhancing or simulating pelvic floor muscle exercises is increasing. In 2019, the POGP reported that there is ‘insufficient evidence to recommend that NMES should be an addition to PFMT’ (POGP, 2019). However, POGP acknowledged that anecdotal evidence suggests NMES to be useful, especially in cases where a patient cannot perform a successful pelvic floor muscle contraction by themselves. The statement from the POGP also reported that there is some empirical evidence to suggest that NMES alone is as useful as pelvic floor muscle training, and can be useful in the management of overactive bladder symptoms (POGP, 2019).
Nurses should be aware that some patients will experience discomfort or pain when using NMES equipment (Chêne et al, 2013).
Conclusion
It is important to recognise that despite a lack of long-term studies providing evidence of efficacy, pelvic floor rehabilitation is a rapidly developing market. Containment with pads is the widely accepted solution for managing incontinence, with many people going on to experience pelvic organ prolapse as a result of their condition going untreated. Instead, treatment should have a larger role in incontinence management, whether this be in the form of reinforced PFMT, or through the use of extra interventions. This could influence the role of the urology nurse in terms of choosing and applying the best course of treatment for patients within urodynamics.
Several clinical studies report NMES of the pelvic floor muscles for rehabilitation to be a safe and effective treatment for incontinence (Sand et al, 1995; Indrekvam and Hunskaar, 2002). However, it is essential to be aware of the cautions and contraindications that may apply when using an electrical stimulation rehabilitation device in treatment.
Purchase the full article from the BJN